As an exceptional functional filler material, the performance of Light (Nano) Calcium Carbonate (CaCO₃) is fundamentally determined by its key technical specifications. These include calcium content, particle size, particle size distribution, and powder morphology. This article delves into the techniques for regulating its morphology and the resultant diverse applications across industries. Achieving the precise particle size and morphology outlined below requires advanced and reliable processing equipment. At EPIC Powder, we specialize in providing the grinding and classifying solutions necessary to meet these industrial demands for calcium carbonate.

1. Key Technologies for Controlling Calcium Carbonate Morphology
The preparation of light (nano) calcium carbonate primarily utilizes carbonation and double decomposition methods. Other techniques like micro-emulsion, liquid membrane, and sol-gel methods are also used, each capable of producing CaCO₃with different crystal morphologies.
Common Preparation Methods for Light (Nano) Calcium Carbonate
A. Carbonation Method
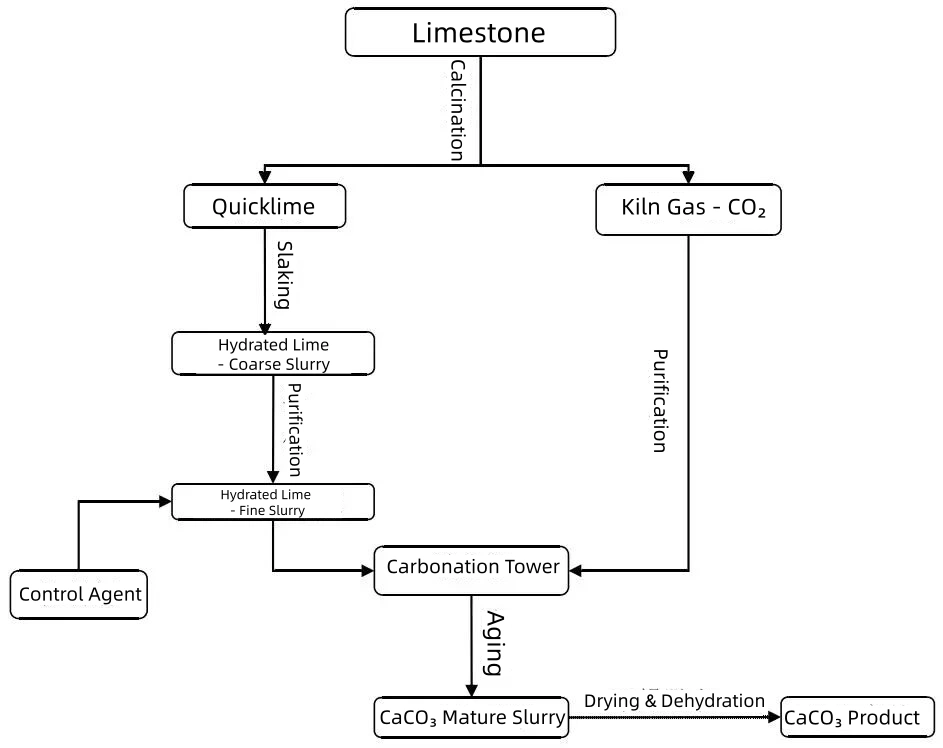
The carbonation method is the primary technique for industrial-scale production, known for its mature technology. Based on different production processes and gas-liquid contact methods, it can be further divided into continuous bubbling, intermittent bubbling, continuous spray, and high-gravity carbonation methods. The main process, as illustrated below, involves calcining limestone to produce quicklime (calcium oxide) and CO₂ gas. The quicklime is then slaked and purified to create a clean Ca(OH)₂ slurry. After adding control agents, this slurry enters a carbonation tower where purified kiln gas (CO₂) is introduced for carbonation. Finally, the mature calcium carbonate slurry undergoes separation, drying, and dehydration to yield the final product.
Flowchart of the Carbonation Process for CaCO₃
| Reaction System | Preparation Method | Advantages | Disadvantages |
| Ca(OH)₂-H₂O-CO₂ Reaction System | Batch Bubbling Carbonation Method | Low cost, simple operation, high production capacity | High energy consumption, uneven product particle size |
| Continuous Spray Carbonation Method | Continuous operation, high production capacity, controllable product | High equipment requirements, high technical content, difficult management | |
| Batch Stirring Carbonation Method | Controllable product, commonly used | High equipment investment, complex operation | |
| High-Gravity Reactive Crystallization Method | Short reaction time, concentrated product particle size range | High requirements for reaction equipment, high energy consumption | |
| Ca²⁺-H₂O-CO₂ Reaction System | Calcium Chloride – Ammonium Carbonate Method | Easily available and low-cost raw materials, simple preparation process operation, high product whiteness | Difficult to remove impurity ions |
| Calcium Chloride – Sodium Bicarbonate Method | |||
| Lime – Sodium Carbonate Method | |||
| Ca²⁺-R-CO₂ Reaction System | Gel Method | Controllable product, suitable for studying crystallization process | Difficult to remove organic matter |
| Microemulsion Method | Prevents product agglomeration, simple operation | Mainly used in experiments |
Comparatively, the carbonation method offers superior control over the crystal form and morphology of calcium carbonate. Crystal formation occurs during the carbonation stage. By precisely controlling process parameters such as Ca²⁺ concentration, carbonation temperature, CO₂ flow rate, pH value, and the use of additives, different product characteristics can be achieved. The main advantages are its low cost and suitability for large-scale production. However, traditional carbonation methods can face challenges like uneven particle size distribution and lower efficiency when producing special morphologies. To address these issues, researchers are continuously exploring innovative carbonation processes, optimizing carbonation tower designs, developing new crystal modifiers, and refining reaction conditions.
B. Double Decomposition Method
This method involves the direct reaction between a soluble calcium salt and a carbonate (or bicarbonate) in a solution under controlled conditions. Depending on the reaction medium, it can be implemented through various techniques such as micro-emulsion, gel, or template methods. The core reaction remains the interaction between Ca²⁺ and CO₃²⁻ ions, typically achieved in systems like Ca²⁺–H₂O–CO₃²⁻ or Ca²⁺–R–CO₃²⁻ (where R represents an organic medium). The key to this method is the use of appropriate control agents to direct the crystal morphology and polymorph.
Flowchart of the Double Decomposition Process
While the double decomposition method can produce spherical calcium carbonate with regular morphology and good dispersion, the raw materials are often more complex and can introduce impurities. This makes it less suitable for large-scale industrial production compared to the carbonation method. Current research is focused on overcoming this bottleneck by using by-product calcium sources like carbide slag, phosphogypsum, and steel slag, coupled with purification processes.
2. Industrial Applications of Different CaCO₃ Morphologies
The unique properties imparted by different morphologies make nano-calcium carbonate suitable for a wide range of specialized applications.
Spherical
Spherical nano-CaCO₃ features a simple structure, small bulk volume, and low oil absorption. It offers excellent smoothness, fluidity, high opacity, and strong ink absorbency. Its primary applications are in paper making, lubricants, and electronic ceramics.
Needle-like (Whisker)
Needle-like nano-CaCO₃, or calcium carbonate whiskers, typically refers to single-crystal fibers with an aspect ratio greater than 10. Their perfect crystal structure provides significantly better reinforcement and toughening effects than common toughening agents. As a reinforcing filler, it markedly improves the strength, elongation, hardness, and wear resistance of materials, especially enhancing the flex resistance of rubber.
Chain-like
Chain-like nano-CaCO₃ is an excellent reinforcing filler for rubber. During the mixing (compounding) process, the chain structure breaks, creating highly active points that bond with the rubber polymer chains. This significantly improves its dispersion within the matrix and greatly enhances its reinforcing effect.
Cubic
With its simple structure, small bulk volume, and good flowability, cubic CaCO₃ provides high opacity, smoothness, and brightness in paper. When added to plastics, it enhances the material’s strength, impact resistance, and processability.
Plate-like
The ability of plate-like particles to layer and compact makes them highly valuable in the paper industry. They greatly increase paper opacity and produce paper with excellent brightness, printability, ink absorption, and smoothness. Due to their high whiteness, moderate oil absorption, and orienting effect in polymer matrices, they are also commonly used in coatings, inks, and plastic films. Studies show that their unique arrangement as fillers and reinforcers provides advantages like high smoothness, gloss, and good mechanical properties, and in some composites, high resistivity and elastic modulus.
Amorphous
Amorphous nano-CaCO₃ boasts an exceptionally high specific surface area (up to 600 m²/cm³), approximately 20 times that of its crystalline counterparts. This makes it highly effective at adsorbing colors and odors, with the ability to release the adsorbed gases under certain conditions. It can also serve as an inexpensive absorbent for toxic metals and as a monodisperse filler for various polymers.
The efficient production and modification of these specialized calcium carbonate grades depend heavily on state-of-the-art machinery. Contact EPIC Powder today to discuss how our ultrafine grinding jet mills and classifying mills can help you optimize your production process for these high-value materials.
Epic Powder
Epic Powder, 20+ years of work experience in the ultrafine powder industry. Actively promote the future development of ultra-fine powder, focusing on crushing, grinding, classifying and modification process of ultra-fine powder. Contact us for a free consultation and customized solutions! Our expert team can provide high-quality products and services to maximize the value of your powder processing. Epic Powder—Your Trusted Powder Processing Expert!

“Thanks for reading. I hope my article helps. Please leave a comment down below. You may also contact EPIC Powder online customer representative Zelda for any further inquiries.”
— Jason Wang, Engineer